恭喜实验室刘启阳(研究生)与赵盼、高贺闻(本科生)的文章被 J Org Chem(中科院2区)接
发布时间:2026-03-14 17:14:43
Visible-Light-Driven Radical Amination/Cyclization of Arylethynyl Quinazolinones to Access Sulfonaminated Quinolino[2,1-b]quinazolines
Qiyang Liu,a Jinwei Yuan,a,*Jianning Zhang,a Pan Zhao,a Hewen Gao,a Liangru Yang,a Ji Ma,b,*Lingbo Quc,d,*
The Journal of Organic Chemistry, 2026, (Accepted)
https://doi.org/10.1021/acs.joc.5c03167
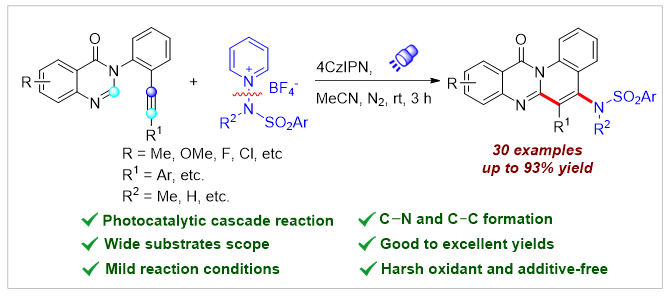
A visible-light-driven radical amination/cyclization of arylethynyl quinazolin-4(3H)-ones was developed using N-aminopyridinium salts as sulfonamino radical precursors. A series of sulfonaminated quinolino[2,1-b]quinazolines were successfully synthesized under oxidant- and additive-free conditions. The transformation features a broad substrate scope, good to excellent yields, and mild reaction conditions. Notably, the amination/cyclization reaction could proceed smoothly under natural sunlight irradiation.
Qiyang Liu,a Jinwei Yuan,a,*Jianning Zhang,a Pan Zhao,a Hewen Gao,a Liangru Yang,a Ji Ma,b,*Lingbo Quc,d,*
The Journal of Organic Chemistry, 2026, (Accepted)
https://doi.org/10.1021/acs.joc.5c03167

A visible-light-driven radical amination/cyclization of arylethynyl quinazolin-4(3H)-ones was developed using N-aminopyridinium salts as sulfonamino radical precursors. A series of sulfonaminated quinolino[2,1-b]quinazolines were successfully synthesized under oxidant- and additive-free conditions. The transformation features a broad substrate scope, good to excellent yields, and mild reaction conditions. Notably, the amination/cyclization reaction could proceed smoothly under natural sunlight irradiation.